| leipner photosynthesis consulting |
leipner photosynthesis consulting |
|
| home - services - applications - background - about lpc | ||
| contact
leipner photosynthesis consulting email: leipner@photosynthesis.ch |
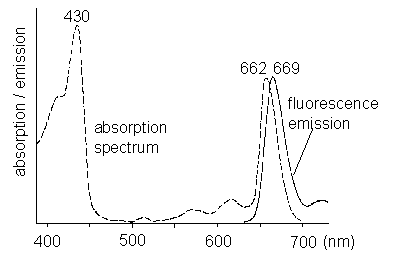
Scientific background Chlorophyll a fluorescence measurements in plant biology PrinciplesLight energy that is absorbed by chlorophyll in a leaf can undergo three fates: a) it can be used to drive photosynthesis (photochemistry), b) it can be dissipated as heat or c) it can be re-emitted as red fluorescence (Figure 1).
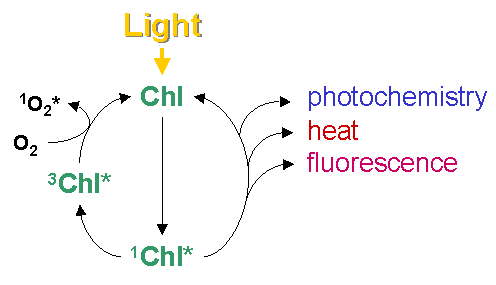
Figure 1: Absorption and emission spectrum of chlorophyll a. These three process occur in competition. Since the sum of rate constants is constant, any increase in the efficiency of one process will result in a decrease in the yield of the other two. Therefore, determining the yield of chlorophyll fluorescence will give information about changes in the efficiency of photochemistry and heat dissipation (Figure 2).
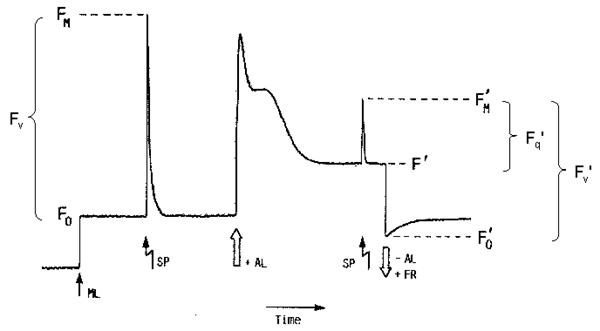
Figure 2: Possible fates of excited chlorophyll How to measure?The most useful and widely used chlorophyll fluorescence technique is the so-called quenching analysis of modulated fluorescence by the saturation pulse method. A typical measurement is shown in Figure 3. A leaf is dark adapted for at least 15 min (depending on temperature) prior to the measurement. Upon the application of a saturating flash (about 8000 μmol m-2 s-1 for 0.6 - 1 s), fluorescence raises from the ground state value (Fo), which is the fluorescence determined in darkness by a weak measuring beam, to its maximum value, namely Fm. This measurement allows the determination of the maximum quantum efficiency of photosystem II (PSII) primary photochemistry, given as Fv/Fm. Upon subsequent application of constant illumination, a transient rise in fluorescence yield is observed. This is due to a lag phase before carbon fixation starts. Whilst electron transport starts within milliseconds upon illumination, carbon fixation needs first to be light-activated. Thereafter, upon the onset of photochemical and heat dissipation processes, the fluorescence yield is quenched and reaches a steady state value (F'). This effect was first observed by Kautsky in 1931. The application of a saturating flash in the presence of actinic light allows the determination of the maximum fluorescence in the light-adapted state (Fm'). A decrease in Fm' as compared to Fm indicates the presence of non-photochemical quenching (NPQ). The difference between Fm' and F' reflects the photochemical part of fluorescence quenching which is expressed in the fluorescence parameter Fq'/Fm', also called Genty parameter. Finally, the minimum fluorescence in the light-adapted state (Fo') can be measured by applying a pulse of far red light. Knowing Fo', the photochemical efficiency factor (Fq'/Fv') and the efficiency of open PSII reaction centres (Fv'/Fm') can be calculated.
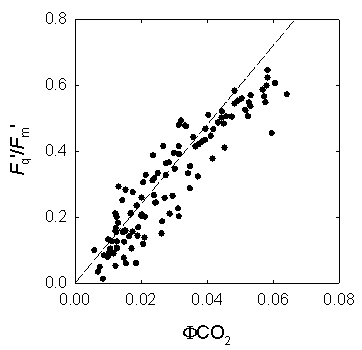
Figure 3: Measurement of chlorophyll fluorescence by the saturation pulse method (adapted from Van Kooten & Snell, 1990). The fluorescence parametersFq'/Fm'Of particular interest in plant physiology are studies of the photosynthetic activity. Photochemical processes can be assessed easily by chlorophyll fluorescence measurements. Perhaps the single most useful fluorescence parameter is the so called Genty parameter. The Genty parameter is calculated as: Fq'/Fm' = (Fm'-F') / Fm' Fq'/Fm' has also been termed φPSII, DeltaF/Fm', (Fm'-Ft)/Fm' and (Fm'-Fs)/Fm' (where DeltaF = Fq', and Ft and Fs equate to F'). A great practical advantage of Fq'/Fm' is the fact that for the calculation of this parameter only Fm' and F' are needed to be determined, both parameters can be measured in the light, i.e. no dark-adapted leaf material is needed. However, due to the fact that Fq'/Fm' is strongly affected by the light intensity precaution has to be done especially during measurements under natural conditions where changes of the incident light intensity hardly can be avoided. Since Fq'/Fm' is theoretically proportional to the operating quantum efficiency of PSII photochemistry (afterwards called PSII quantum efficiency), as such, it is a measure of the proportion of the light absorbed by photosystem II that is used in photochemistry. The PSII quantum efficiency is affected by the level of electron acceptors, usually NADP+, available at the acceptor side of PSI. Consequently, Fq'/Fm' decreases in situations with limiting consumption of NADPH like for example at low internal CO2 concentration. Changes in Fq'/Fm' can be attributable to differences in the capacity for electron flux on the reducing side of PSII, as mentioned above, but also to down-regulation of PSII, since Fq'/Fm' is the product of the PSII efficiency factor (Fq'/Fv') and the PSII maximum efficiency (Fv'/Fm') which is affected by antenna quenching. The PSII quantum efficiency frequently exhibits a strong, quantitative relationship with the quantum yield of CO2 assimilation (φCO2). Since the theoretical minimum of consumed electrons per CO2 fixed is 4, the ratio of Fq'/Fm' to φCO2 would be 8:1, which is also the theoretical minimum quantum requirement for non-cyclic electron flow per CO2 fixed. This is true for many C3 plants, but not for C4 plants. In maize, the Fq'/Fm':φCO2 ratio is about 12:1 probably due to a lower PSII/PSI ratio; this means that 12 photons are required to fix one molecule CO2 (Figure 4). Furthermore, the linear relationship between Fq'/Fm' and φCO2 is broken when photorespiration, Mehler reaction or other alternative electron sinks to CO2 fixation are engaged. Simultaneous determination of Fq'/Fm' by chlorophyll fluorescence measurements and of φCO2 or φO2 by gas-exchange measurements allows to quantify these alternative pathways.
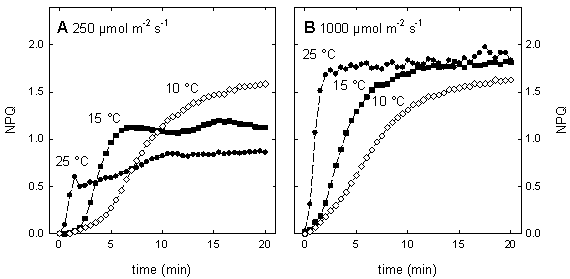
Figure 4: The relation between quantum efficiency of PSII (Fq'/Fm') and quantum efficiency of CO2 fixation (φCO2) in maize leaves grown in the field. ETRMultiplied with the amount of absorbed light by PSII, Fq'/Fm' is a measure of the rate of linear electron transport through photosystem II. The electron transport rate (ETR) can be calculated as: ETR = Fq'/Fm' × PPFD × aL × (PSII/PSI) where PPFD is the photosynthetic photon flux density (in μmol quanta m-2 s-1), aL the leaf absorption, and PSII/PSI equates to the ratio between light absorption of photosystem II and I. Whilst Fq'/Fm', PPFD and aL can be measured without difficulties using a fluorometer, light sensor and integrating sphere, the stoichometry of the photosystems can only be assumed resulting often in questionable values of ETR. However, within species grown at identical conditions measurements of ETR are reasonable. Calculating the electron transport rate (ETR) reveals that at low light intensity ETR increases linearly with light intensity until a certain level where electron transport cannot keep up with the absorbed light energy, respectively when photosynthesis is not light limited anymore. The maximum ETR is achieved when the capacity of the sum of all electron sinks (carbon fixation, photorespiration, nitrate assimilation, Mehler reaction) is reached. Fq'/Fv'The PSII efficiency factor (Fq'/Fv'), which is mathematically the same as the widely used coefficient of photochemical quenching (qP), is often thought to be a proxy of the redox state of the primary electron acceptor QA. It must be emphasised here that this is true only if there were little or no connectivity among PSII centres. Rather, changes in Fq'/Fv' reflect differences in the capacity for photochemistry at PSII. The PSII efficiency factor is calculated as: Fq'/Fv' = (Fm'-F') / (Fm'-Fo') NPQExcess of excitation energy can be de-excited by thermal dissipation processes. Non-photochemical quenching of chlorophyll fluorescence is an indicative of the level of non-radiative energy dissipation in the light-harvesting antenna of photosystem II. The importance of the non-photochemical quenching results from the fact that it shows that the level of excitation energy in the PSII antenna can be regulated. This is thought to prevent over-reduction of the electron transfer chain and, therefore, provides protection from photodamage. The most straightforward way to quantify non-photochemical quenching is by measuring the fluorescence parameter NPQ, which is calculated as: NPQ = (Fm/Fm') - 1 The parameter NPQ is derived from the Stern-Volmer equation and can be used to follow changes in apparent quencher concentration. NPQ is related to the rate constant for excitation quenching by regulated thermal dissipation (k'N). Non-photochemical quenching is induced under conditions when the photosynthetic apparatus cannot use the total of absorbed light energy for photochemistry, which can occur already at quite low light intensity even under optimum conditions for photosynthesis. Stress conditions such as high light intensity, low internal CO2 concentration due to drought or low temperature markedly promote non-photochemical quenching. Therefore, the amount of non-photochemical quenching is an indicator of the stress severity, respectively of the excess of absorbed light. At moderate light intensity, the NPQ steady state value is dependent from the temperature. However, at a certain point NPQ cannot increase further, illustrated by the temperature independence of the steady state level of NPQ at high light intensity (Fig. 5). This maximum value of NPQ reflects the capacity of quencher, which can be altered by acclimation or adaptation.
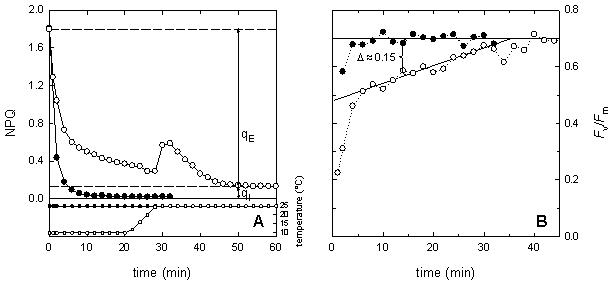
Figure 5: Temperature dependence of the induction of non-photochemical quenching (NPQ) in maize leaves at moderate light intensity (A, 250 μmol m-2 s-1) and at high light intensity (B, 1000 μmol m-2 s-1). Plants were grown at 25°C under a PPFD of 350 μmol m-2 s-1 The kinetics of NPQ development is strongly temperature dependent (Fig. 5). Low temperature decreases the rate of NPQ development irrespectively of the light intensity. Bilger and Björkman (1991) demonstrated that the development of NPQ upon exposure of leaves to excess light is, at least partially, determined by the rate of zeaxanthin formation, which is slowed down at low temperature due to the temperature dependence of the enzyme violaxanthin de-epoxidase. Zeaxanthin determines how rapidly the quenching develops. In contrast, the rate of the zeaxanthin-independent part of NPQ is little affected by temperature. Further information about NPQ can be obtained by studying the relaxation of NPQ in darkness. In higher plants, non-photochemical quenching can be divided into at least two different components according to their relaxation kinetics. The most rapidly relaxing component is the DeltapH- or energy-dependent non-photochemical quenching, often named qE. The level of qE is dependent upon both the DeltapH, which is essential, and zeaxanthin content that acts as a positive effector. Although, the dark relaxation of qE is determined by the disappearance of the DeltapH it is noticeably temperature dependent. As seen in Figure 6, qE relaxes considerably slower at 10°C as compared to 25°C. The NPQ sustaining at low temperature can be rapidly reversed upon warming or by uncoupling of the thylakoid membrane with nigericin. The dark-sustained, low-temperature-stimulated NPQ is apparently directly dependent on lumen acidification and chloroplastic ATP hydrolysis. The remaining part of NPQ that relaxation is markedly slower than that of qE is called photoinhibitory non-photochemical quenching, qI. The contribution of both kinds of non-photochemical quenching can be very different in leaves exhibiting the same values for NPQ (Fig. 6). Increasing proportion of qI on NPQ is an indicator for enhanced stress.
Figure 6: Temperature dependence of the dark relaxation of NPQ (A) and of Fv/Fm (B) in maize leaves at 25°C (filled symbols) and at 10°C (open symbols). After 20 min of relaxation at 10°C, temperature was increased to 25°C (see inlet in A). NPQ was induced by high light intensity (1200 μmol m-2 s-1) for 20 minutes at the given temperature, 25°C and 10°C, respectively. Fv'/Fm'An other useful parameter to describe energy dissipation is Fv'/Fm' which provides an estimate of the PSII quantum efficiency if all PSII centres were in the 'open' state at the point of measurement. Its value is largely determined by down-regulation, which appears to involve the operation of one or more processes that increase the rate constant for non-radiative decay of excitation energy within the pigment matrix associated with PSII. The quantum efficiency of open PSII reaction centres is calculated as: Fv'/Fm' = (Fm'-Fo') / Fm' Even though the determination of Fo' is needed to calculate Fv'/Fm', the advantage of this parameter is that no dark-adapted leaf material is needed. Values of NPQ can be difficult to interpret in leaves with reduced Fm, for example, in a leaf that was already photoinhibited and exhibited therefore a lower Fm. Fv/FmThe reaction centre of photosystem II is frequently undergoing photoinduced damages of the D1 protein in a process referred to as photoinhibition. Photoinhibition is induced by inoptimal electron transfer as it occurs enhanced during stress conditions when the capacity of photochemistry and dissipative mechanisms is exhausted. PSII can tolerate an increase in its inactivation to quite some extent, but it can do so only up to the capacity of its repair system. As soon as the capacity for repair of damaged PSII centres is exceeded, an irreversible inhibition of PSII can be detected in vivo as a decrease in the chlorophyll fluorescence ratio Fv/Fm which is a measure of the maximum quantum efficiency of photosystem II primary photochemistry. The maximum quantum efficiency of PSII photochemistry is calculated as: Fv/Fm = (Fm-Fo) / Fm A decrease in Fv/Fm usually is originated by a decrease in Fm in combination with an increase in Fo. The latter is provoked by dissociation of light harvesting pigment system of PSII from the PSII core. It is discussed that decrease of Fv/Fm might be a mix of photoprotection and photodamage. Stress can affect Fv/Fm by enhanced damage of the D1 protein and / or by reduced activity of the D1 repair system. The D1 protein repair can be studied by convolution of recovery kinetics of Fv/Fm. In particular, the C-terminal processing of the precursor D1 protein in the repair of PSII reaction centre is temperature-dependent. Another factor, which affects the recovery kinetics of Fv/Fm, is light. In comparison to complete darkness, low light intensities enhance the D1 protein repair. The parameter Fv/Fm is very useful to estimate the extent of photoinhibition of photosynthesis. However, in some circumstances the precise determination can be erroneous, especially, when sustainable non-photochemical quenching is feigning a decrease in Fv/Fm. This can happen if dark adaptation is conducted at low temperature and Fv/Fm determined too early. Photoinhibition, respectively low Fv/Fm, usually affects other chlorophyll fluorescence parameter. As mentioned above, the capacity of NPQ might be lowered in photoinhibited level due to a low value of Fm. Since photoinhibition will reduce the electron pressure on the reducing site of PSI, these leaves are often characterised by higher values of Fq'/Fv'.
|
further
information ... about the application of chlorophyll a fluorescence in plant biology can be found in: Fracheboud Y. & Leipner J. (2003) The application of chlorophyll fluorescence to study light, temperature, and drought stress. In Practical Applications of Chlorophyll Fluorescence in Plant Biology (eds J.R. DeEll & P.M.A. Toivonen), pp 125-150. Kluwer Academic Publishers, Dordrecht. |
| © 2010 jl |